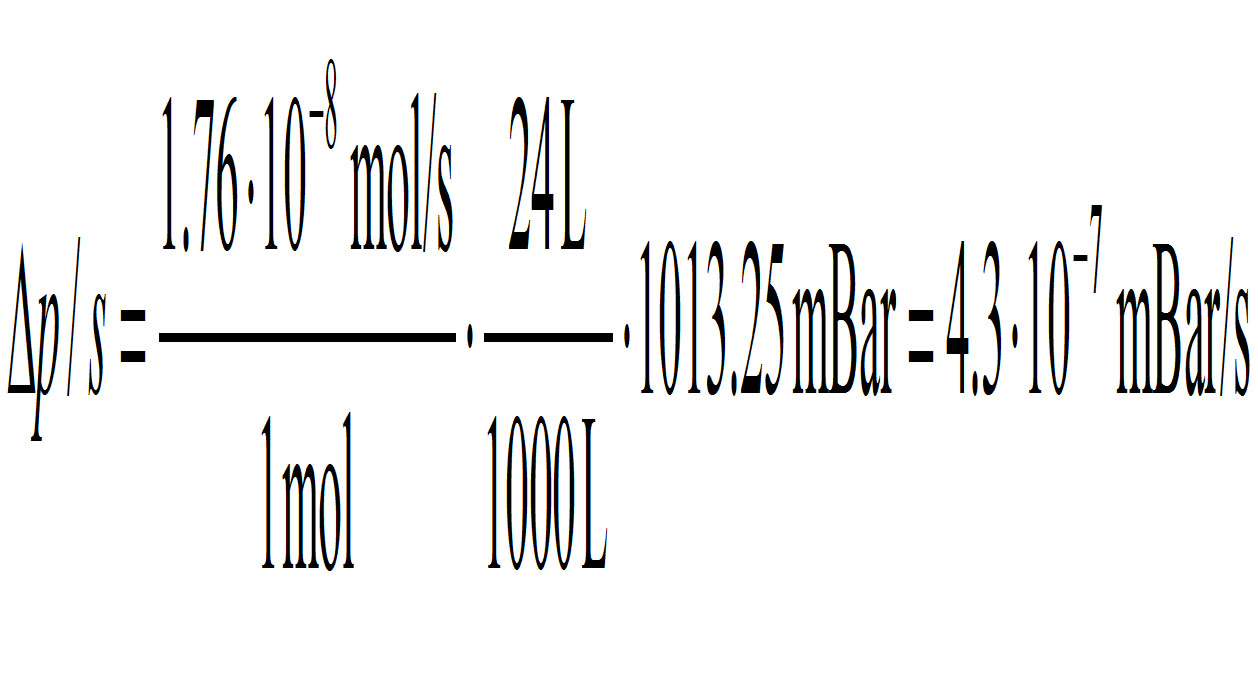MetalicaRap:Physics Principles
Principles
Following EBM metal vaporisation loss of vacuum leading to loss of beam quality
cal
Problem: Pressure from evaporated material
Example from “Practial manufacturing walk through” will add in soon!
- Vaporizing Titanium at with .04 W into metal volume evaporating at the price of <math>5*10^4 J/cm^3</math> evaporates
<math>\frac{0.04W}{5*10^4 J/cm^3}=8*10^{-7} cm^3</math>
With the density of metallic Ti 4.5 g/cm3 the above volume amounts to 3.6x10-6 g. And with a molar mass of Ti 204.37 g/mol it is 1.76x10-8 mol. Now: ONE mole of a gas takes a volume of 24 Liter at a pressure of 1 atmosphere or 101,325 Pa = 1013.25 millibar. Relative to this an amount of 1.76x10-8 mol vaporized titanium in a 1000 L (1 m3) volume will pr second contribute to the pressure with the following value
<math>=\Delta P /S =\frac{1.76*10^{-8} mol/S}{1 mol}*\frac{24L}{1000L}*1013.25 mBar =4.43*10^{-7} mBar</math>
Thus the pump has to remove 4.3x10-7 millibars of pressure per second. Maybe this is not at all a problem. I don’t know about pumping capacities. I just wanted to put attention to it.
PS: According to my tables the heat of evaporation of Ti is not 5x104 but 4x104 J/cm3 (8.953 MJ/kg times 4.5x103 kg/m3
Discussion
[1] To improve layout of above? <math>2*10^4</math>